From creating new antimicrobials to combating Alzheimer’s disease
Structural biology provides insights into the diverse functions of fibrous protein in humans, amphibians, and bacteria
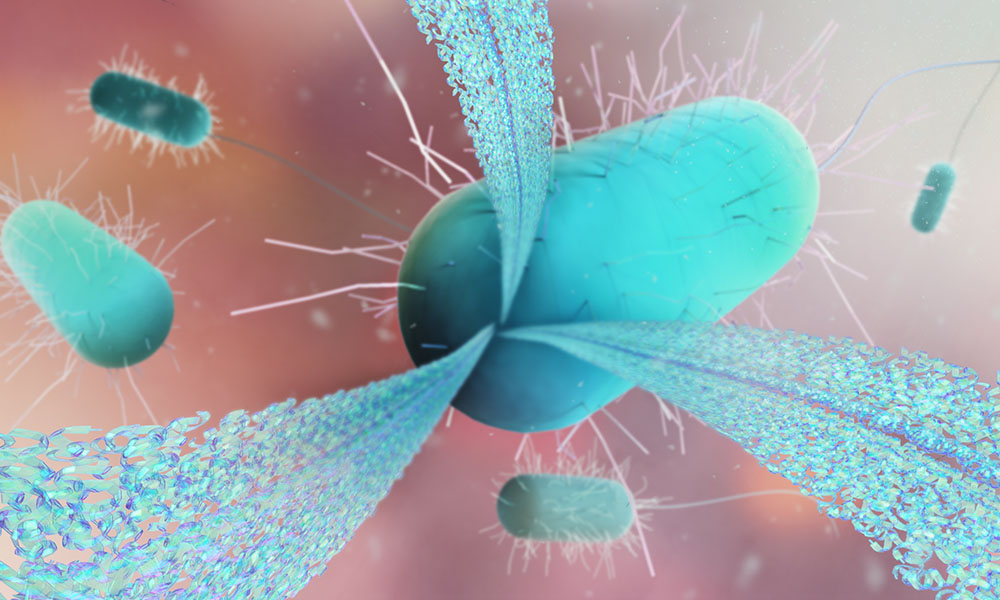
What do Alzheimer’s disease, dental cavities, and a blocked sink drain have in common? They can all involve nanometre-sized bacterial protein fibres that can form molecular constructions as strong as steel.
These fibres form a scaffold that stabilises extremely sturdy layers of bacteria on various surfaces, such as human teeth, a catheter, and even the inside of the sink drain. The fibres can probably also form in the human body during infection, killing immune cells and making bacterial infections more aggressive.
The Landau Group at EMBL Hamburg and at Technion – Israel Institute of Technology discovered the structural basis of these bacterial fibres’ stability and activity. The findings present new ways to develop therapeutic strategies that treat infections not by killing the bacteria but by disrupting the fibres that serve as their ‘weapons’. The aim is to stop bacteria from doing harm while the host fights the infection on its own.
This approach offers a major advantage: in contrast to antibiotics, anti-fibre drugs might be less prone to antibiotic resistance, which the World Health Organization considers one of the top 10 global public health threats. If it is not tackled, antibiotic resistance could lead to diseases such as tuberculosis and pneumonia becoming completely untreatable. In many countries, certain antibiotics are already ineffective in more than half of patients, and the number of drug-resistant bacterial species is growing. This research could help to find an urgently needed alternative to antibiotics.
Certain proteins have a fascinating ability to spontaneously self-assemble into supramolecular fibres, thereby gaining new properties. With the use of structural biology techniques, the Landau Group analysed the atomic structure of fibres secreted by certain bacteria in the gut and in many foods. They saw that these structures have striking similarities to a group of human proteins called amyloids, which also form supramolecular structures. Many amyloids, for instance amyloid-beta and alpha-synuclein, are linked to disease. They can lump into plaques in the brain and have been found in people with Alzheimer’s, Parkinson’s, and several other neurodegenerative diseases.
“This connection between bacterial fibres in the gut and human amyloids in the brain could help us find a factor contributing to the propensity for Alzheimer’s and Parkinson’s diseases,” said Group Leader Meytal Landau. “It’s possible that certain bacteria present in the gut secrete fibres that are transmitted to the brain and trigger human amyloids to aggregate. This suggests a similar mechanism to mad cow disease, which is caused by eating meat contaminated with infectious proteins called prions.”

Interestingly, the same amyloids associated with neurodegeneration also have a protective role in fighting microbial threats. The Landau Group discovered that human and amphibian peptides (small proteins) that are used to combat bacteria also form fibres. These peptides self-assemble into unique fibrous nanostructures, which in the presence of bacteria restructure to become a deadly antimicrobial weapon. Such fibres could serve as a durable, active coating for medical devices or implants, industrial equipment, food packaging, and more.
Landau joined EMBL Hamburg in mid-2019 as visiting group leader. At EMBL, she has access to a synchrotron for crystallographic studies and to cryo-electron microscopes for high-resolution structural studies – tools not available to her group at the Technion in Israel.
“The EMBL beamlines are among the few across Europe suited for crystallographic studies of amyloid fibrils,” she explained. “EMBL Hamburg is very attractive because of its integrated facility for structural biology and access to diverse techniques under one roof.”